London Hosts the Third International Summit on Human Genome Editing
/Scientists from around the world gathered last week at the Francis Crick Institute for the Third International Summit on Human Genome Editing to discuss the rapid advances in the field and the ethical implications involved.
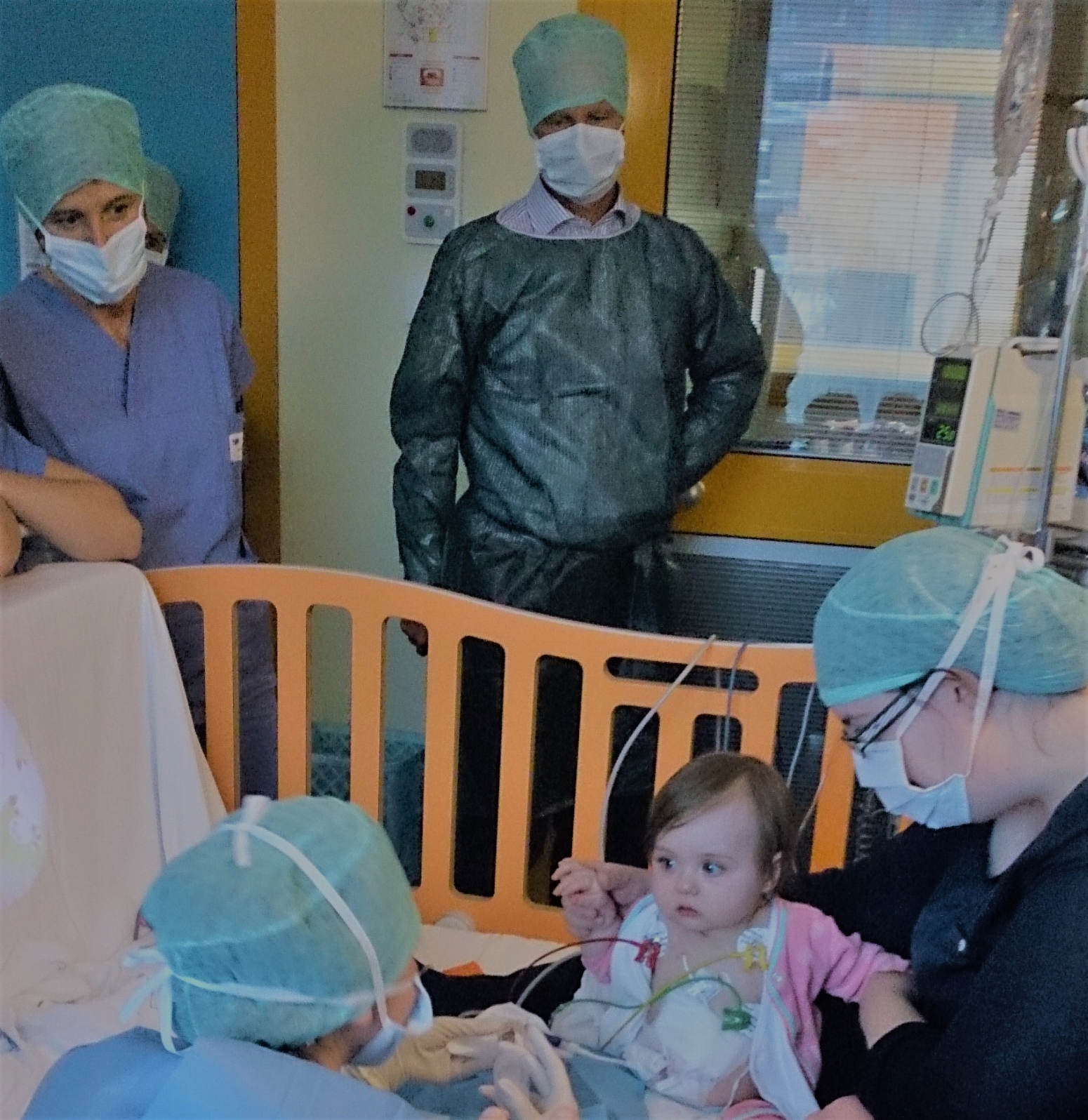
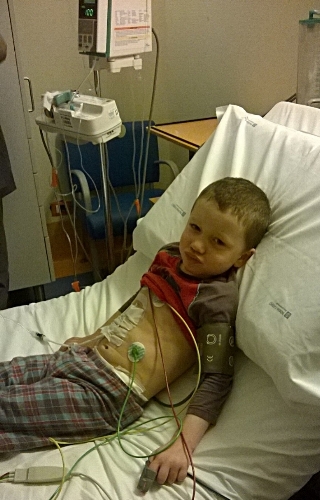
The successful trial of patients with sickle cell disease and the progress of several other human trials using CRISPR in somatic cells (those belonging to a single person) to treat inherited diseases was celebrated. However, the revelation that it may “technically” be possible to edit germline cells (embryo cells that get inherited by future generations) to treat niche genetic diseases within five years, highlighted the controversial ethical issuessurrounding this technology which is racing ahead of society at large.
Without more concrete international regulation, there is little to stop this science being developed for human enhancement and nefarious purposes, which would change human DNA, and ultimately, the human race forever.
“Heritable human genome editing remains unacceptable at this time,” key scientists at the concluding meeting announced in a statement issued on 8 March. Failures and concerning results of tests on embryos conducted in the lab added to the mantra of heed and not speed. “Preclinical evidence for the safety and efficacy of heritable human genome editing has not been established, nor has societal discussion and policy debate been concluded.”
Another key issued discussed was the prohibitively expensive price-tag of these drugs (upwards of $2 million) and how to make them affordable and available to those who need them, like those in African communities where sickle cell disease is most prevalent.
Below, is my selection of some of the best of what was published this week, which further explores the most pertinent topics discussed at the summit.
Forthcoming genetic therapies raise serious ethical questions, experts warn - The Guardian
We need rules before we let the genome-editing genie out of the bottle - Financial Times
Why CRISPR babies are still too risky — embryo studies highlight challenges - Nature
Forget designer babies. Here's how CRISPRis really changing lives - MIT Technology Review
Beyond CRISPR babies: How human genome editing is moving on after scandal - Nature
How genome editing will change humanity - The Economist
UK government urged to consider changing law to allow gene editing of embryos - The Guardian
CRISPR technology modifies cells to fight cancer: 'Ultimate tool for manipulating life' - FOX News